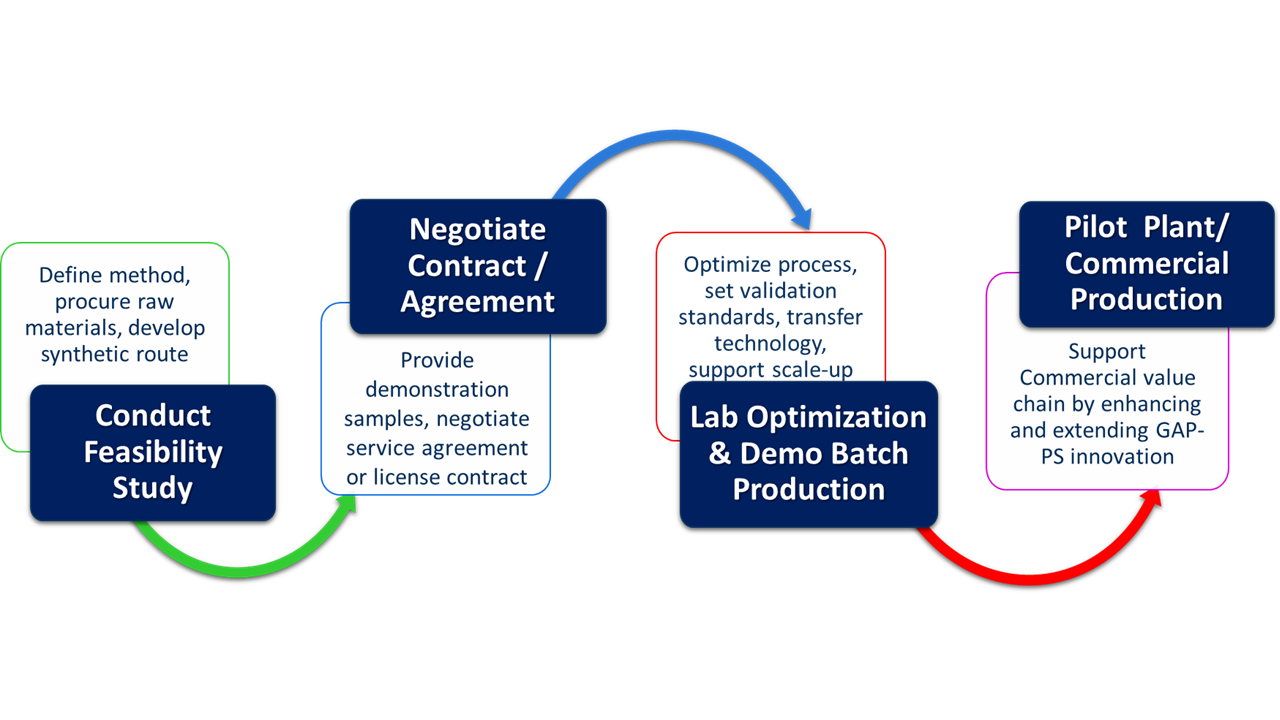
GAP Peptides’ Service Model
Improving Synthesis Efficiency and Economics with Scalable, Sustainable, GAP-PS Chemistry
Collaboration for Acceleration
We utilize a customer collaboration model – listening carefully to critical needs and applying science to develop innovative, sustainable solutions. We find ways to partner and collaborate with our clients that strengthen their competitive position while extending the strategic value of GAP-PS.
Focused on delivering Market growth for our clients, GAP-PS technology provides novel, green synthesis routes that reduce costs, minimize hazardous waste, and provide flexible, easy scaleup.
Proof of Concept Studies
For many developers and manufacturers, innovation is key to their growth strategy. Innovation is at the heart of GAP Peptides.
We deliver practical innovation: working with our clients to develop and optimize green and highly economical routes for their peptide targets. We also provide the technical expertise to support optimization for scaleup projects.
Process Innovation Delivered
GAP Peptides technology provides opportunities to deliver products to customers much faster and at greater scale than traditional synthesis methods. Please contact us to discuss your challenges.